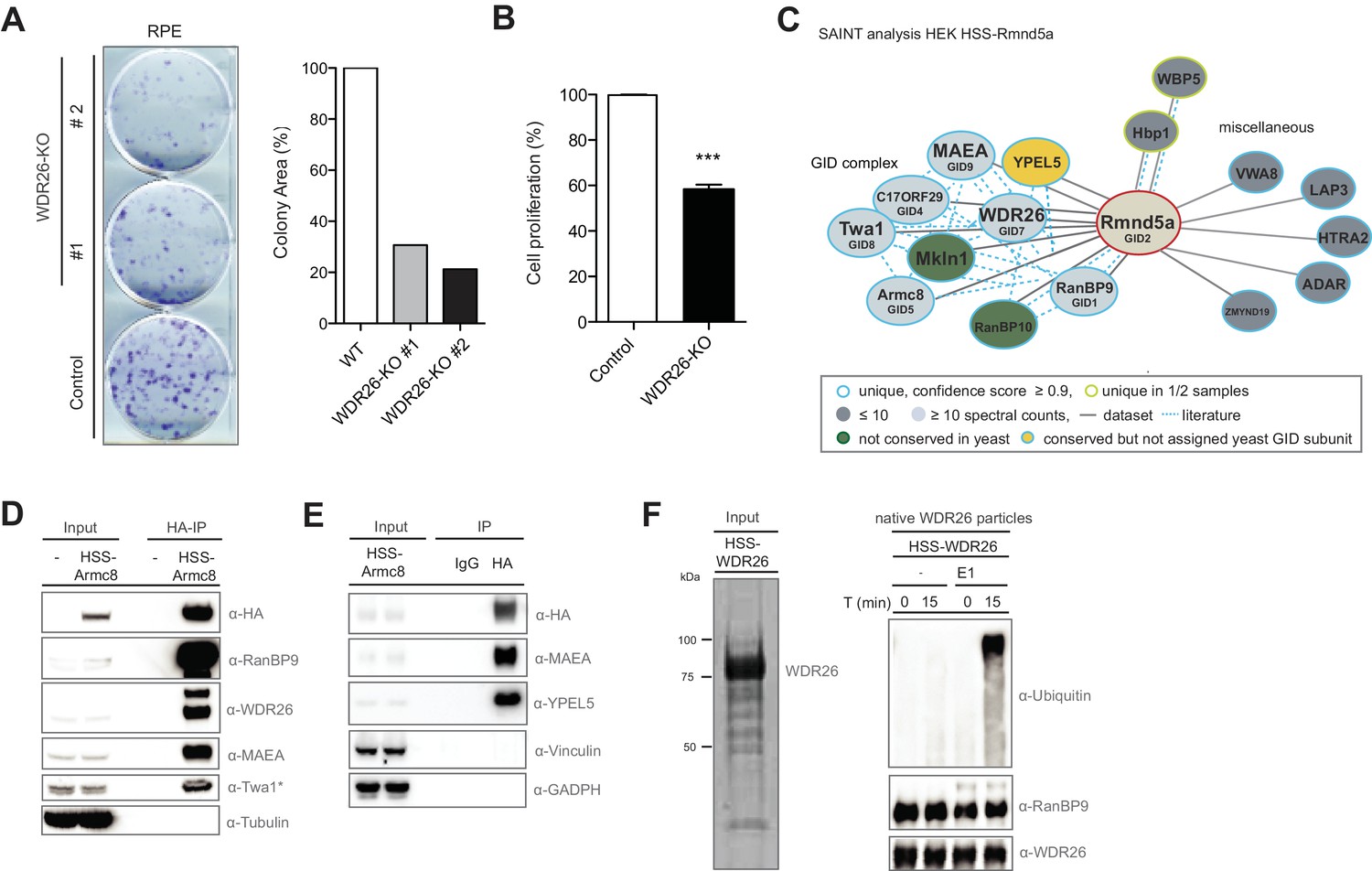
Cryo-EM structures of Gid12-bound GID E3 reveal steric blockade as a mechanism inhibiting substrate ubiquitylation | Nature Communications
GitHub - KratosMultiphysics/GiDInterface: The graphical user interface of Kratos for GiD. Featuring CFD, CSM, DEM, PFEM, etc